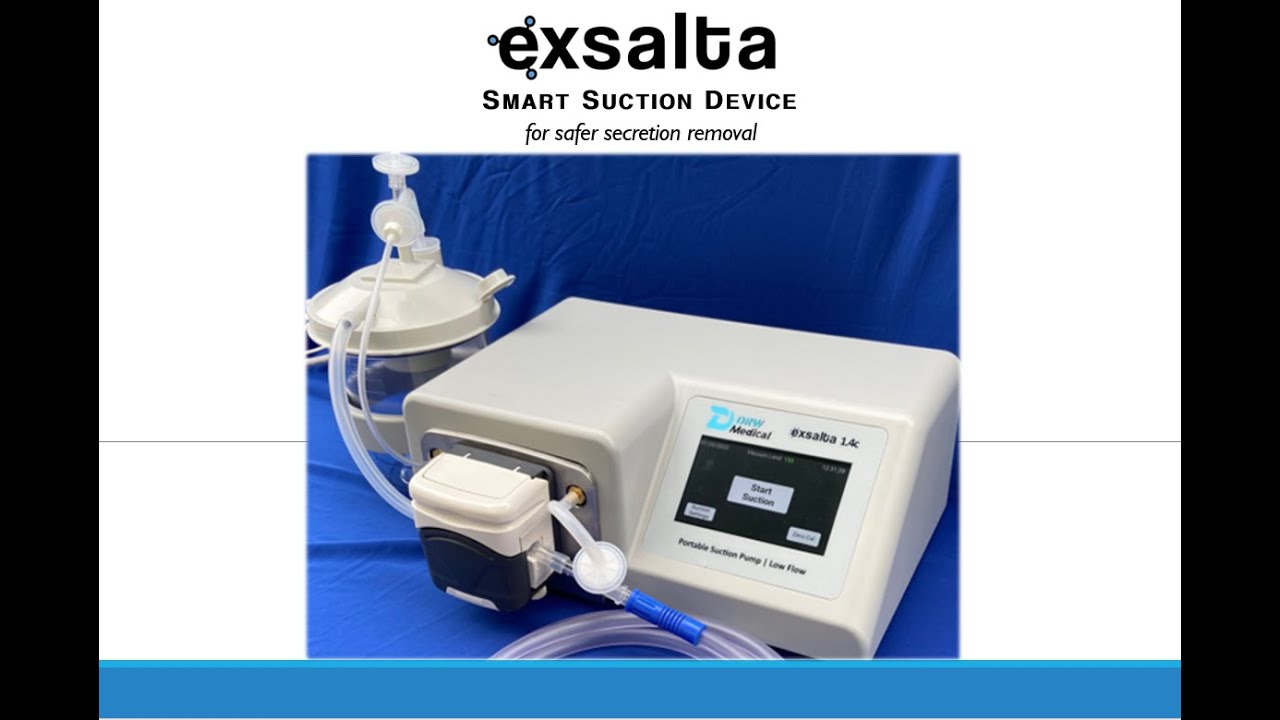
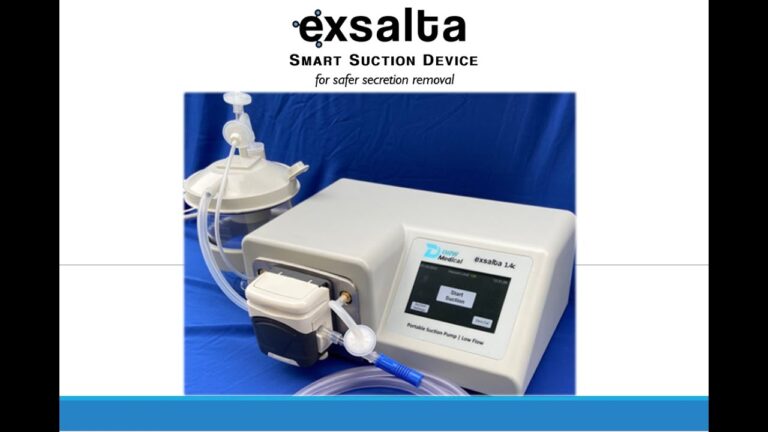
Aston PA., September 16, 2022 – DRW Medical LLC announced today that the U.S. Food and Drug Administration has cleared the Exsalta device for use on patients as indicated. Exsalta is the first peristaltic suction device for clearing endotracheal tube secretions for patients on ventilators. Its exceptional low flow technology, available at any suction setting, reduces risk of negative pressures in the lungs to help prevent alveolar collapse and hypoxia. Exsalta is a smart secretion clearance solution for patients in whom the standard wall suctioning procedure may create adverse events.
The EXSALTA offers clinicians Smart suction technology to help mitigate adverse events associated with standard suctioning techniques.
- Maintain O2 Sat while clearing the ET Tube
- Leave the oxygen for the patient to breathe
- Increase the vacuum strength, not the flowrate
- Smart suction is Low-Flow operation with all the strength of wall suction